China’s Medical Device Listing Is No Longer a One-Time Filing
Since January 2025, over ten provinces have launched listing data governance actions. Classification code mismatch is now the third active suspension trigger, and the one most companies do not track
China’s medical device listing system is no longer a one-time registration. On 17 April 2026, Zhejiang province’s drug and device procurement centre published notice 11330000MB18470516/2026-03450, suspending seven medical device products from online trading due to classification code non-compliance. Three weeks earlier, Shenzhen’s public resources trading centre issued a separate notice requiring non-selected IOL and sports medicine products to adjust listing prices to comply with provincial price ceilings, with products failing to respond within the objection window facing delisting. These are not isolated events: since January 2025, over ten provinces, including Hebei, Shandong, Shaanxi, Hunan, Heilongjiang, Jiangxi and Hubei, have initiated listing data governance actions. China’s device listing system is transitioning from a one-time registration to an active, ongoing compliance obligation.
What Changed, and Why It Matters
The policy-execution gap here is straightforward: most manufacturers entered China’s provincial listing platforms when their products launched and have not revisited those filings since. The regulatory assumption, ‘once listed, always listed’, is no longer valid. Listing status is now subject to active monitoring, price comparison across provinces, and classification code validation. Failure on any of these dimensions triggers suspension.
Three Conditions That Now Trigger Suspension
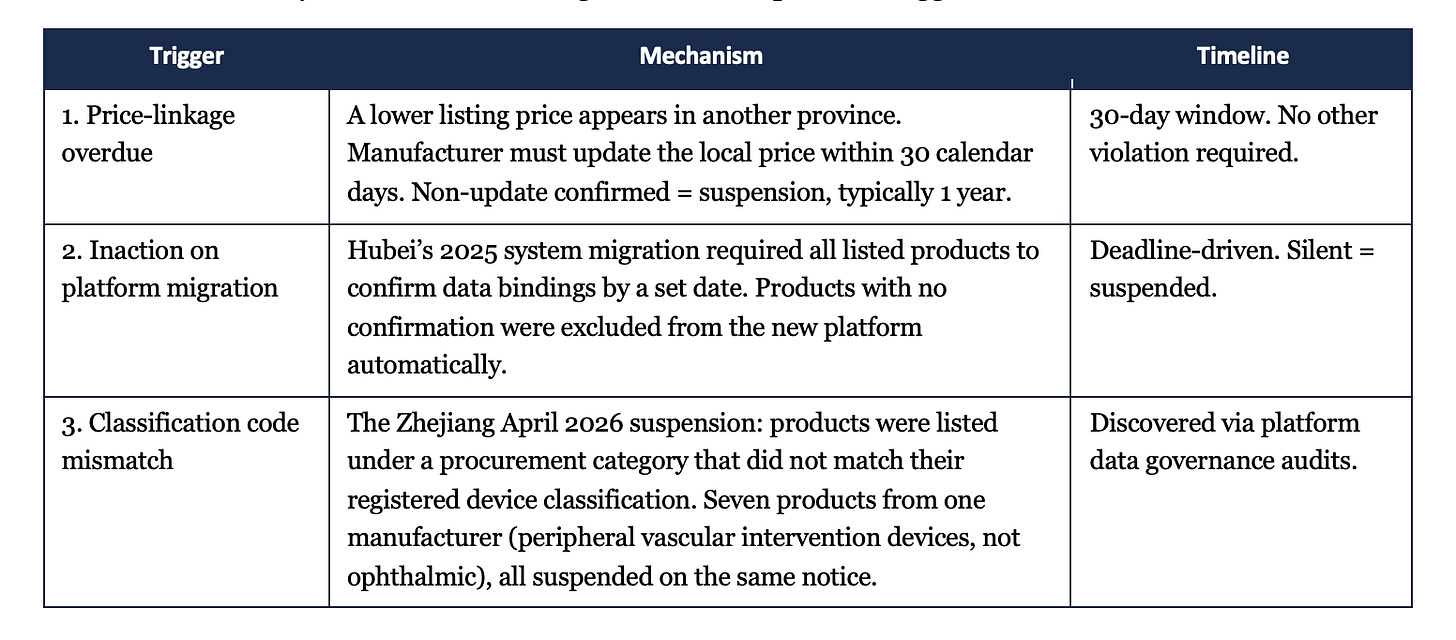
Provincial rules vary in detail but converge on three suspension triggers:
1. Price-linkage overdue
A lower listing price appears in another province. The manufacturer must update the local price within 30 calendar days. Non-update confirmed = suspension, typically 1 year.
30-day window. No other violation required.
2. Inaction on platform migration
Hubei’s 2025 system migration required all listed products to confirm data bindings by a set date. Products with no confirmation were excluded from the new platform automatically.
Deadline-driven. Silent = suspended.
3. Classification code mismatch
The Zhejiang April 2026 suspension: products were listed under a procurement category that did not match their registered device classification. Seven products from one manufacturer (peripheral vascular intervention devices, not ophthalmic), all suspended on the same notice.
Discovered via platform data governance audits.
The Zhejiang case involved peripheral vascular intervention devices, not ophthalmic products. Its significance for IOL manufacturers is structural: it demonstrates that classification code mismatch is now an active suspension trigger alongside the two previously observed mechanisms. The enforcement infrastructure does not distinguish by therapeutic area.
Zhejiang adds a fourth dimension: category-wide exposure. Under Zhejiang’s price-linkage rules, if a manufacturer has six or more products in a single category with non-compliant linkage filings, the entire category is suspended, not just the specific products. For manufacturers with broad product portfolios, one compliance gap can remove an entire device line from the province’s procurement platform simultaneously.
Why This Is Not Indirect for IOL Manufacturers
A common assumption is that high-end intraocular lenses, multifocal, EDOF, toric premium, are largely insulated from listing compliance issues because their principal revenue comes from out-of-pocket patients rather than insurance reimbursement. This assumption is incorrect in a specific, consequential way.
Listing is not a precondition for insurance payment. It is a precondition for any public hospital procurement. A product that is not entered into a province’s healthcare procurement sub-system cannot be ordered by a public hospital, regardless of whether the patient intends to pay out of pocket. In clinical practice, this means a surgeon has no pathway to recommend a product to a patient if it does not appear in the hospital’s billing catalogue, not because of clinical judgment, but because the administrative channel does not exist.
This is not theoretical. In ophthalmic clinical practice before national VBP implementation, the standard workflow was as follows: before a physician could present any IOL option to a patient, whether single-focus, toric, or multifocal, the product had to be entered into the hospital’s billing catalogue. A product absent from that system could not be priced, could not be consent-documented, and could not be ordered. The physician’s judgment about what was clinically appropriate was constrained entirely by what appeared in that catalogue. A listing suspension does not reduce a product’s visibility to surgeons. It removes it.
Zhejiang’s medical security bureau has built real-time digital monitoring of IOL procurement execution across its public hospitals, publishing compliance rates at the institutional level. The infrastructure for detecting listing anomalies is already operating. The April 2026 suspension is a routine output of that infrastructure, not an exceptional enforcement action.
The Precedent: What Happened to Zeiss and an IOL Import Agent
Two cases define the outer boundary of this risk. In October 2025, the national high-value device joint procurement office cancelled Carl Zeiss Meditec AG’s VBP selection status for its bifocal non-toric IOL product (registration number: 国械注进20153161147) after a quality inspection found the product’s spectral transmittance did not meet the required standard. The suspension cascaded across multiple provinces, including Shanghai, Zhejiang, Guangdong, Ningxia and Guangxi. The lesson: listing status is not product-level, it is enterprise-level. A quality or compliance event affecting one SKU generates platform-wide risk flags for the brand.
In early 2023, an IOL import agent, Hangzhou Xiehe Medical Supplies Co., Ltd (杭州协合医疗用品有限公司), acting as the domestic representative for an Indonesian manufacturer’s IOL product, voluntarily withdrew from a Shanghai municipal IOL procurement after winning the bid at RMB 989 per lens. Under Shanghai’s rules, this triggered a two-year suspension of all that company’s IOL products from Shanghai procurement and listing applications, effective February 2023 to February 2025. One decision: two years of market exclusion across an entire product category.
OphthalLogix Assessment: Who Is Exposed, and What to Do
Listing compliance rules are not drafted to disadvantage foreign manufacturers specifically. But their practical impact is uneven based on execution capacity, not intent.
Manufacturers with dedicated, China-based market access teams that actively monitor provincial price databases, track platform migration deadlines, and audit product classification codes are relatively advantaged as smaller or less-resourced competitors are cleared from platforms. The governance trend is accelerating the consolidation of market access capacity at established players.
The countervailing risk: large multinational manufacturers with broad product portfolios face disproportionate exposure to Zhejiang’s category-wide suspension rule. A multi-SKU ophthalmic portfolio is precisely the structure most likely to carry the volume of non-compliant linkage filings that trigger a category suspension. Size and breadth are not a shield here; they increase the surface area for compliance gaps.
The self-audit window is now. Zhejiang’s April 2026 public notice closes on 23 April. If your products are listed in Zhejiang, the question of whether any of them fall under the categories subject to active governance is answerable in minutes on the provincial platform, and the cost of discovering a problem now is a fraction of the cost of receiving a suspension notice.
Key Implications
China’s medical device listing system shifted in 2025–2026 from passive registration to active compliance maintenance. Manufacturers affected are all entities with products on provincial procurement platforms, both foreign and domestic, but the practical burden falls heaviest on those without dedicated market access monitoring. For IOL manufacturers specifically, the listing-suspension risk is not indirect: a suspended product disappears from the hospital procurement channel regardless of whether the end patient is paying out of pocket. The immediate action is a platform audit of active listing status, price-linkage compliance, and product classification accuracy across key provinces, prioritising Zhejiang and Guangdong, given their demonstrated enforcement activity in April 2026.
This content is for informational purposes only and does not constitute legal, regulatory, investment, or medical advice. China’s healthcare policy environment moves quickly; the status of any regulatory development should be verified independently before informing a commercial or compliance decision. OphthalLogix Intelligence accepts no liability for decisions made in reliance on this content.
— The OphthalLogix Intelligence Team